You are using an out of date browser. It may not display this or other websites correctly.
You should upgrade or use an alternative browser.
You should upgrade or use an alternative browser.
How does lower pH improved the efficacy of SO2?
- Thread starter NorCal
- Start date

Help Support Winemaking Talk - Winemaking Forum:
This site may earn a commission from merchant affiliate
links, including eBay, Amazon, and others.
Johny99
Junior Member
- Joined
- Sep 10, 2010
- Messages
- 969
- Reaction score
- 639
Per Mr. Margalit
SO 2 chemistry in aqueous solution
The equilibrium of sulfur-dioxide in water is carried on in two steps:
SO 2 + H 2 O <=======> H + + HSO 3 ;
HSO 3 <=====> H + + SO 3 =
The first one is an equilibrium between molecular SO 2 and bisulfite ion (HSO 3 ), and the second one is between the bisulfite ion and sulfite (SO 3 = ). At lower pH range where the first equilibrium is dominant, the dissociation constant K a of the reaction is defined as: K a = [H + ] x [HSO 3 ] / [SO 2 ] Where: [SO 2 ] ; [HSO 3 ] ; [H+] are the molecular SO 2 ; bisulfite ion; and hydrogen ion concentrations respectively. And because the value pK a is formally defined as: pK a = logK a ; and also pH = log[H + ] ; ====> pK a = logK a = log[H + ]x[HSO 3 ] / [SO 2 ] = log[H + ] log{[HSO 3 ] / [SO 2 ]} = pH log{[HSO 3 ] / [SO 2 ]} ; therefore: which means that the ratio of the bisulfite form (HSO 3 ) to the molecular form [SO 2 ] of sulfur-dioxide, is pH dependent (!)
Simple heh
SO 2 chemistry in aqueous solution
The equilibrium of sulfur-dioxide in water is carried on in two steps:
SO 2 + H 2 O <=======> H + + HSO 3 ;
HSO 3 <=====> H + + SO 3 =
The first one is an equilibrium between molecular SO 2 and bisulfite ion (HSO 3 ), and the second one is between the bisulfite ion and sulfite (SO 3 = ). At lower pH range where the first equilibrium is dominant, the dissociation constant K a of the reaction is defined as: K a = [H + ] x [HSO 3 ] / [SO 2 ] Where: [SO 2 ] ; [HSO 3 ] ; [H+] are the molecular SO 2 ; bisulfite ion; and hydrogen ion concentrations respectively. And because the value pK a is formally defined as: pK a = logK a ; and also pH = log[H + ] ; ====> pK a = logK a = log[H + ]x[HSO 3 ] / [SO 2 ] = log[H + ] log{[HSO 3 ] / [SO 2 ]} = pH log{[HSO 3 ] / [SO 2 ]} ; therefore: which means that the ratio of the bisulfite form (HSO 3 ) to the molecular form [SO 2 ] of sulfur-dioxide, is pH dependent (!)
Simple heh

Boatboy24
No longer a newbie, but still clueless.
I always assumed it was the acid protecting the wine more effectively, thereby requiring less SO2. But that was just an assumption. I think we need @ibglowin to chime in on this one.
Thanks @Johny99, while I cannot distill that paragraph down to a few sentences that I understand, my takeaway is that there actually is a real chemistry reason that SO2 is more effective at lower pH.
Furthermore, here is my understanding of what protects the wine from oxidation and spoilage microbes.
1. Acidity
2. Tannins
3. Alcohol
4. Free SO2
5. Temperature
6. Sanitized contact
7. Vessel that avoids O2 contact
8. Time
Clearly if you do not do a good job with #6 and #7, there is little you can do with the other factors. Of all the remaining, having the proper pH and keeping an eye on SO2 will protect most wines in most environments.
So, what if your pH is 4.1 and any further adjustments, even .5 g/l of tartaric, makes your wine undrinkable? Oh yea, this is after you have blended it with all your lower pH wine. Can you really stress the other areas that protect the wine to compensate for your number one defense effective SO2? Is there any otter choice?
Furthermore, here is my understanding of what protects the wine from oxidation and spoilage microbes.
1. Acidity
2. Tannins
3. Alcohol
4. Free SO2
5. Temperature
6. Sanitized contact
7. Vessel that avoids O2 contact
8. Time
Clearly if you do not do a good job with #6 and #7, there is little you can do with the other factors. Of all the remaining, having the proper pH and keeping an eye on SO2 will protect most wines in most environments.
So, what if your pH is 4.1 and any further adjustments, even .5 g/l of tartaric, makes your wine undrinkable? Oh yea, this is after you have blended it with all your lower pH wine. Can you really stress the other areas that protect the wine to compensate for your number one defense effective SO2? Is there any otter choice?
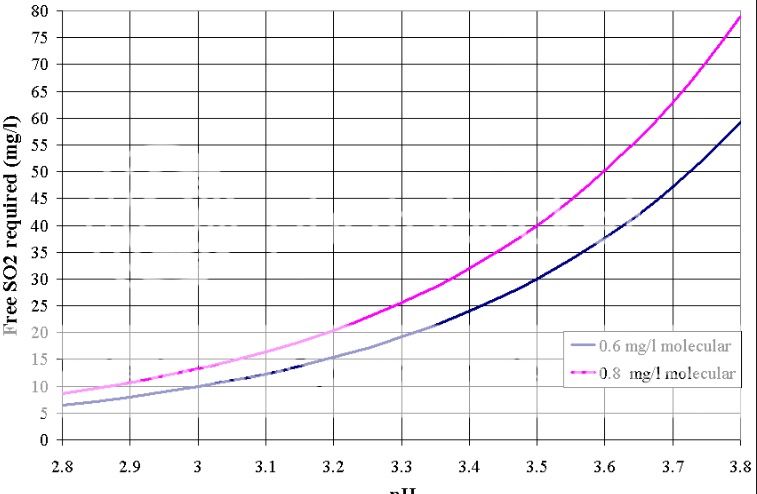
Sulfur dioxide in wines exists in two different chemical forms, SO2 and HSO3(-1). These two chemical species are called “molecular SO2” and “bisulfite,” respectively.
When you measure Free SO2 you measure both the molecular SO2 and the bisulfite, but only the molecular SO2 is effective as a bacterial preservative. And in wines, only 1% to 7% of the Free SO2 is present as molecular SO2.
These two chemical forms are related in the following chemical equation:
SO2 + H2O = HSO3(-1) + H(+1)
What this equation says is that if you have more acid, which is the same as having a low pH wine, then you get a higher percentage of the molecular SO2. If you have less acid, or a high pH wine, you get a much lower percentage of the molecular SO2. The following table shows the percent of the Free SO2 that is present as molecular SO2 for different pH levels in wine.
What the table says is that you need 9.45 times as much Free SO2 if your wine has a pH of 4.0 as you would if the same wine had a pH of 3.0.

When you measure Free SO2 you measure both the molecular SO2 and the bisulfite, but only the molecular SO2 is effective as a bacterial preservative. And in wines, only 1% to 7% of the Free SO2 is present as molecular SO2.
These two chemical forms are related in the following chemical equation:
SO2 + H2O = HSO3(-1) + H(+1)
What this equation says is that if you have more acid, which is the same as having a low pH wine, then you get a higher percentage of the molecular SO2. If you have less acid, or a high pH wine, you get a much lower percentage of the molecular SO2. The following table shows the percent of the Free SO2 that is present as molecular SO2 for different pH levels in wine.
What the table says is that you need 9.45 times as much Free SO2 if your wine has a pH of 4.0 as you would if the same wine had a pH of 3.0.

Johny99
Junior Member
- Joined
- Sep 10, 2010
- Messages
- 969
- Reaction score
- 639
Thanks @Johny99, while I cannot distill that paragraph down to a few sentences that I understand, my takeaway is that there actually is a real chemistry reason that SO2 is more effective at lower pH.
So, what if your pH is 4.1 and any further adjustments, even .5 g/l of tartaric, makes your wine undrinkable? Oh yea, this is after you have blended it with all your lower pH wine. Can you really stress the other areas that protect the wine to compensate for your number one defense effective SO2? Is there any otter choice?
Yes, it is chemistry and Mike did a nice job of it.
 . I was too lazy to use my own words.
. I was too lazy to use my own words. If you have a high pH wine, and don't want to lower it, often for good reason as you note, you should just be more careful. Sulphate to the level you feel comfortable with. For some that is zero. Keep as clean as you can, and a bit of prayer to the wine gods doesn't hurt. I wouldn't expect it to last too long, so drink it early and enjoy. Better to drink it young than make it undrinkable.
stickman
Veteran Winemaker
- Joined
- Jun 16, 2014
- Messages
- 1,930
- Reaction score
- 2,248
NorCal, in the past I've made some acid adjustments that caused the wine to taste too tart during the bench trial, but once the wine was adjusted and aged a few months in the cellar, and dropped potassium in the form of tartrates, it tasted great. You may want to try adjusting a split and chill proofing in the fridge to see how that tastes (if you haven't already).
Johny99
Junior Member
- Joined
- Sep 10, 2010
- Messages
- 969
- Reaction score
- 639
Thank God there is a chart.
Or wine maker mags calculator. https://winemakermag.com/1301-sulfite-calculator
Cludgy sometimes, but easy to use.
Similar threads
- Replies
- 9
- Views
- 425
- Replies
- 16
- Views
- 457
- Replies
- 2
- Views
- 996